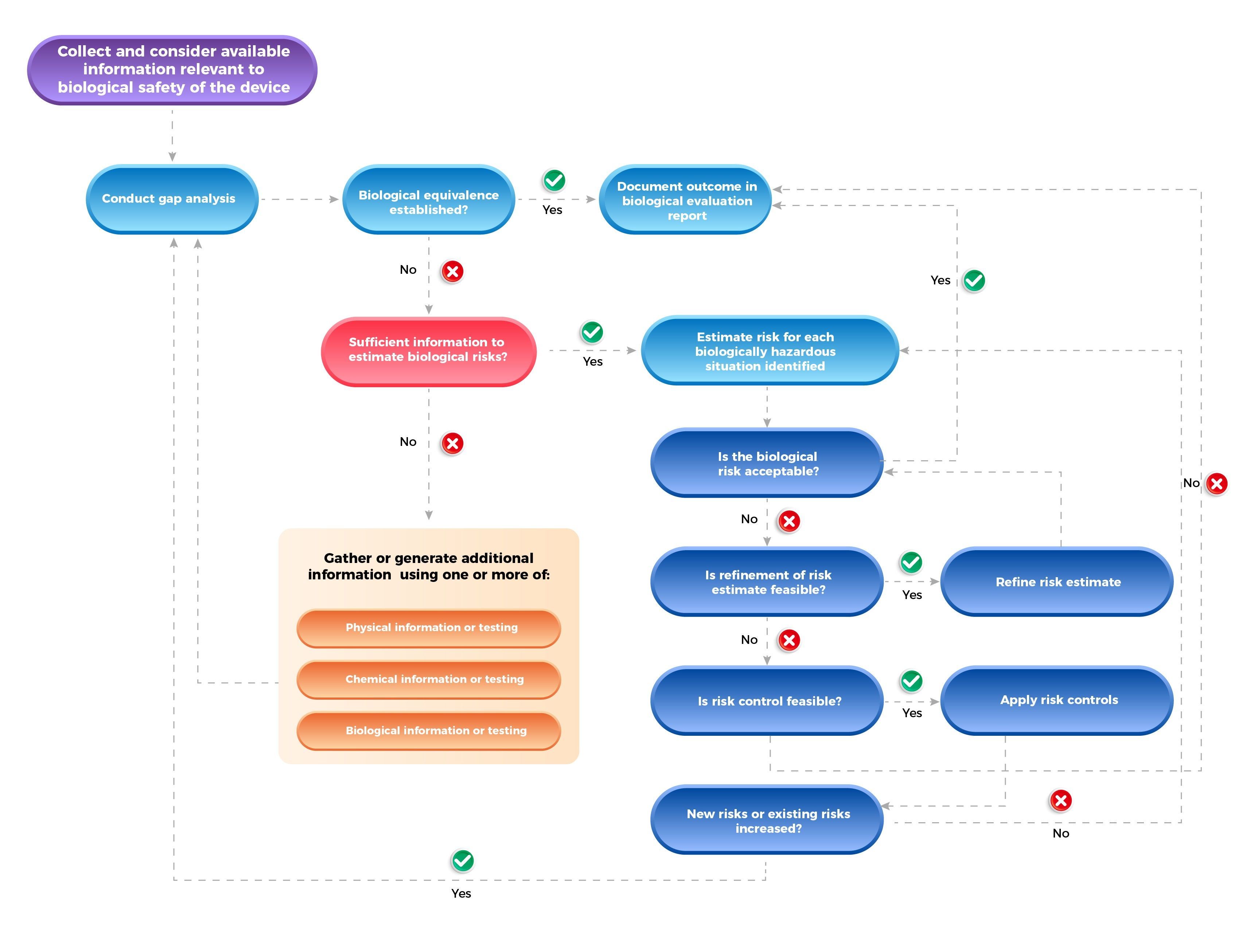
Biological Evaluation within a Risk Management Framework
 Medical device configuration and listing of a medical device’s materials of construction (qualitative) and where necessary, the proportion and amount (mass) of each material in the medical device (quantitative)
Medical device configuration and listing of a medical device’s materials of construction (qualitative) and where necessary, the proportion and amount (mass) of each material in the medical device (quantitative) The physical and chemical characteristics of the various materials of construction and their composition
The physical and chemical characteristics of the various materials of construction and their composition Any history of clinical use or human exposure data
Any history of clinical use or human exposure data Any existing toxicology and other biological safety data on product and component materials, breakdown products and metabolites
Any existing toxicology and other biological safety data on product and component materials, breakdown products and metabolites Test procedure
Test procedureBiological Evaluation Pathways
 Chemical Characterization
Chemical Characterization
 Toxicological Risk Assessment
Toxicological Risk Assessment
 Biological Testing Plan
Biological Testing Plan
 Biocompatibility Testing
Biocompatibility Testing
 Biological Evaluation
Biological Evaluation Chemical Characterization
Chemical Characterization
 Toxicological Risk Assessment
Toxicological Risk Assessment
 Biological Testing Plan
Biological Testing Plan
 Biocompatibility Testing
Biocompatibility Testing
 Biological Evaluation
Biological EvaluationChemical Characterization
 Access to a database of over 6,000 extractables and experience testing over 4,500 leachables, supporting comprehensive chemical characterization.
Access to a database of over 6,000 extractables and experience testing over 4,500 leachables, supporting comprehensive chemical characterization. Over 10 years of experience in chemical characterization for global medical device submissions.
Over 10 years of experience in chemical characterization for global medical device submissions. Professional structure elucidation team supporting accurate identification and analysis of device materials.
Professional structure elucidation team supporting accurate identification and analysis of device materials. Expertise in leachables testing and mass spectrometry for reliable data generation.
Expertise in leachables testing and mass spectrometry for reliable data generation. Guaranteed data integrity to meet national and international regulatory requirements.
Guaranteed data integrity to meet national and international regulatory requirements.
 10+
10+




Biocompatibility Testing
 GLP-, ISO 17025-, and ASCA-accredited laboratory, supporting globally recognized quality systems and regulatory compliance.
GLP-, ISO 17025-, and ASCA-accredited laboratory, supporting globally recognized quality systems and regulatory compliance. Comprehensive in vivo and in vitro testing, including cytotoxicity, irritation, sensitization, systemic toxicity, pyrogenicity, hemocompatibility, genotoxicity, and implantation studies.
Comprehensive in vivo and in vitro testing, including cytotoxicity, irritation, sensitization, systemic toxicity, pyrogenicity, hemocompatibility, genotoxicity, and implantation studies. Conformance with the latest ISO 10993 standards.
Conformance with the latest ISO 10993 standards. Experienced pathologists and toxicologists supporting accurate interpretation and reporting.
Experienced pathologists and toxicologists supporting accurate interpretation and reporting. Over 270,000 biocompatibility studies conducted, providing robust historical data for regulatory submissions.
Over 270,000 biocompatibility studies conducted, providing robust historical data for regulatory submissions.Toxicological Risk Assessment and Biological Evaluation

Over 38,000 historical toxicological assessments.

Experience from over 2,000 programs.

Use of internationally recognised toxicity prediction software and data sources.

Experienced team of toxicologists.

High-quality reports supporting global regulatory submissions.

Over 38,000 historical toxicological assessments.

Experience from over 2,000 programs.

Use of internationally recognised toxicity prediction software and data sources.

Experienced team of toxicologists.

High-quality reports supporting global regulatory submissions.


